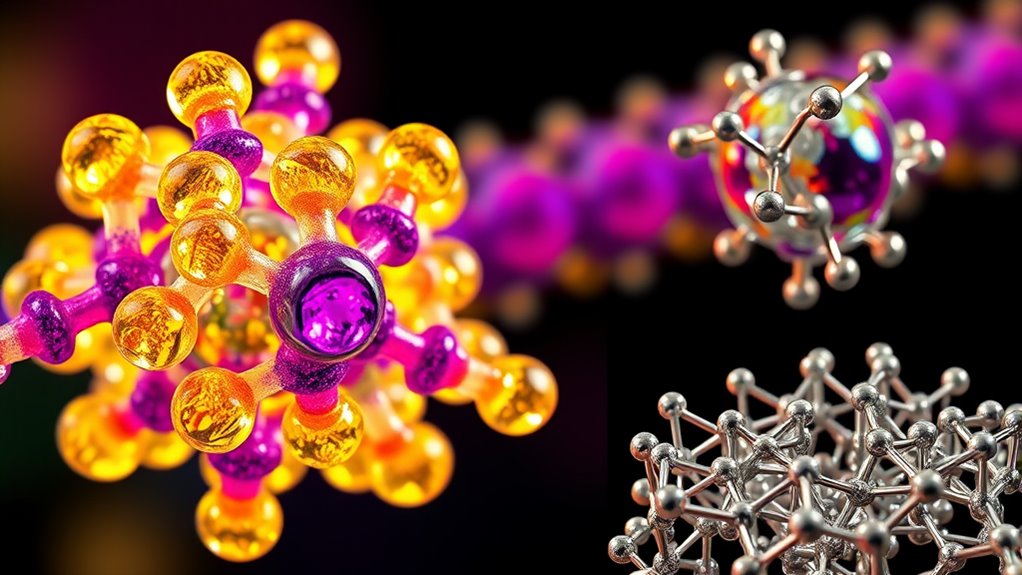
Chemical bonding involves three main types: ionic, covalent, and metallic bonds. Ionic bonds occur when electrons transfer from one atom to another, creating charged ions that attract each other. Covalent bonds form when atoms share electrons, shaping molecules’ structures and properties. Metallic bonds feature delocalized electrons that allow metals to conduct electricity and be shaped easily. Exploring these bond types helps you understand how substances form and behave, offering a deeper view into material properties.
Key Takeaways
- Ionic bonds involve electron transfer, forming charged ions and strong electrostatic attractions, common in metal-nonmetal compounds.
- Covalent bonds occur through electron sharing between atoms, creating molecules with varying bond strengths and shapes.
- Metallic bonds feature a ‘sea of electrons,’ allowing metals to conduct electricity and exhibit malleability.
- Bond type influences material properties such as melting point, reactivity, and physical state.
- Understanding these bonds explains the formation, structure, and behavior of different chemical substances.

Have you ever wondered what holds atoms together to form everything around us? The answer lies in the way atoms interact through chemical bonds. These bonds are the forces that keep atoms connected, creating molecules and solids with unique properties. The most common types are ionic, covalent, and metallic bonds, each with distinct characteristics resulting from how atoms share or transfer electrons. Understanding the types of bonds is essential to grasp the diversity of material properties in chemistry. Focusing on covalent bonds, these form when atoms share electrons through electron sharing. Unlike ionic bonds, where electrons transfer from one atom to another, covalent bonds involve a mutual sharing of electrons to achieve stability. This sharing fills the outer electron shells, making atoms more stable and creating molecules. The bond types in covalent bonding can vary depending on how many pairs of electrons are shared. For example, a single bond involves sharing one pair of electrons, a double bond shares two pairs, and a triple bond shares three. These different bond types influence the strength, length, and reactivity of molecules. For instance, single bonds are generally longer and weaker than double or triple bonds, which are shorter and stronger. This electron sharing defines many properties of molecules, determining their shape, polarity, and how they interact with other substances.
In ionic bonds, instead of sharing electrons, atoms transfer electrons from one to another, leading to the formation of charged ions. This transfer creates electrostatic attractions between positively charged cations and negatively charged anions, resulting in an ionic bond. These bonds typically form between metals and nonmetals. The resulting compounds tend to be crystalline solids with high melting points because the electrostatic forces are strong and hold the ions tightly together. Ionic bonds are responsible for the structure of common salts like sodium chloride.
Metallic bonds are a different story. They involve a ‘sea of electrons’ that are delocalized across a lattice of metal atoms. These free-moving electrons allow metals to conduct electricity and heat efficiently. Metallic bonds give metals their malleability and ductility because the electron cloud can slide past the metal ions without breaking the bond. This electron sharing in metallic bonds is different from covalent bonds because the electrons are not associated with any specific atom, but instead belong to the entire metal structure.
Understanding these bond types and the role of electron sharing helps explain how different substances form and why they have the properties they do. Whether atoms transfer or share electrons, the nature of their bonds shapes the entire world around us.

LINKTOR Chemistry Molecular Model Kit (444 Pieces), Student or Teacher Set for Organic and Inorganic Chemistry Learning, Motivate Enthusiasm for Learning and Raising Space Imagination, A Fullerene Set
FOR BASIC TEACHING TO ADVANCED SCIENCE: 444 pieces molecular model kit, including 136 atoms, 158 bonds and 150…
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
Frequently Asked Questions
How Do Intermolecular Forces Influence Chemical Bonding?
Intermolecular forces, like van der Waals and hydrogen bonds, play a vital role in chemical bonding by affecting how molecules interact and stick together. You’ll notice that stronger forces, such as hydrogen bonds, cause substances to have higher boiling points and greater stability. These forces influence physical properties and determine how molecules organize, ultimately shaping the behavior and characteristics of materials you encounter daily.
Can a Molecule Have Both Ionic and Covalent Bonds?
Yes, a molecule can have both ionic and covalent bonds. For example, in a compound like sodium chloride, sodium forms ionic bonds with chloride, but within the chloride ion, covalent bonds exist between atoms. Molecular hybridization helps explain these structures, and bond energy comparison reveals that ionic bonds generally have higher energy than covalent ones. This combination influences the molecule’s properties, making it unique and versatile in various chemical reactions.
What Role Do Electrons Play in Metallic Bonding?
Imagine electrons as the social butterflies of metallic bonding, gracefully mingling in the electron sea. They become delocalized electrons, freely moving around metal atoms and creating a strong, cohesive bond. These electrons act like a flexible glue, allowing metals to bend and conduct electricity. By roaming freely, they give metals their characteristic properties, making them both durable and excellent conductors, all thanks to these lively, delocalized electrons.
How Does Bond Polarity Affect Molecule Properties?
Bond polarity markedly influences molecular properties like boiling point, solubility, and reactivity. When a bond is polar, electrons are unevenly shared, creating partial charges that lead to stronger intermolecular forces. This makes polar molecules more likely to dissolve in water and have higher boiling points. Conversely, nonpolar bonds result in weaker forces, affecting how molecules interact. Understanding bond polarity helps predict a molecule’s behavior in different environments.
Are There Bonds Stronger Than Ionic or Covalent?
Yes, bonds can be stronger than ionic or covalent ones. For example, nuclear bonds, like those in atomic nuclei, have incredible bond strength and stability, far surpassing chemical bonds. These bonds hold protons and neutrons together with immense force. Additionally, some covalent bonds, especially triple bonds, are extremely strong and stable. So, depending on the context, there are bonds with higher bond strength and greater bond stability than typical ionic or covalent bonds.
ionic and covalent bond educational toys
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
Conclusion
Understanding chemical bonds is like learning the threads that weave the fabric of matter. Ionic, covalent, and metallic bonds each play a unique role, shaping the properties of everything around you. When you grasp these bonds, you unseal a universe of possibilities, much like a master weaver creating intricate patterns. Remember, these bonds are the silent architects of the world, holding everything together in a delicate, yet powerful, dance of attraction.
metallic bonding demonstration kit
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.

Old Nobby Organic Chemistry Set (425 pc) Molecular Model Kit with Atoms, Bonds, Guide, Bond Removal Tool – STEM Science Kits for Kids Toys Chemistry Set for Students Teachers Young Scientists
Comprehensive Molecular Learning Kit: Includes high-quality color-coded atoms and bonds for constructing 3D models of compounds; ideal for…
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.